Polymer in
Medicine
CE 435
Introduction to Polymers
Department of Chemical Engineering
University at Buffalo-SUNY
Jeremy Robinson
Pierre M. Saint Louis
Anoop Padmaraju
Submitted:
04/03/01
Table of Contents
|
Introduction |
2 |
|
A brief history
of polymers in medicine |
4 |
|
Cellophane |
6 |
|
PGA, PLA, and
PLGA |
9 |
|
Polydimethyl
siloxane |
12 |
|
Polyethylene and
PMMA |
15 |
|
Polytatrafluoroethylene |
16 |
|
Polyurethane |
19 |
|
Conclusion |
22 |
|
References |
23 |
Introduction
Biomaterials are substances
other than food or drugs contained in therapeutic or diagnostic systems that
are in contact with tissue or biological fluids. They are used in many
pharmaceutical preparations, for example, as coatings for tablets or capsules
or as components of transdermal patches. Biomaterials play a central role in
extra Corporeal devices, from contact lenses to kidney dialyses, and are
essential components of implants,
from
vascular grafts to cardiac pacemakers1.
Like their counterparts of long ago,
medical practitioners today often seek to cure ailments or improve a patient’s
quality of life by replacing a defective body part with a substitute. But until
quite recently, physicians were limited to using off-the-shelf supplies that
weren’t designed for the application. Motivated by a need for custom-made
materials for specific medical applications, materials scientists, chemists,
chemical engineers, and researchers in other disciplines have turned their
attention to creating high-performance biomaterials. Among the new crop of
substances are novel biodegradable polymers and modified natural substances
designed for use in a wide range of implantable applications including
orthopedic and dental devices, drug-delivery systems, tissue engineering
scaffolds, and other uses1.
Biodegradable polymers take center stage
in a great variety of research efforts. Materials that can decompose and
disappear from the body are desirable for short-term applications in
orthopedics, tissue engineering, and other areas, where, for example, a
physician may need a device to hold a bone in place long enough for the body to
heal. They are many current biomaterials applications found in about 2,700
different kinds of medical devices, 2,500 separate diagnostic products, and
39,000 different pharmaceutical preparations2.
Table 1 |
|||
|
|
Applications of Biomaterials2 |
|
|
|
|
|
|
|
|
Polymer |
Applications |
Polymer |
Applications |
|
PDMS |
Catheters,
heart |
Polytetrafluoroe |
Heart valves |
|
|
Valves |
thylene |
Vascular grafts |
|
|
|
|
Nerve repair |
|
Polyurethane |
ventricular assist |
Polyethylene |
Catheters, hip |
|
|
Devices |
|
prostheses |
|
|
|
Polymethylmetha |
Fracture
fixation |
|
PGA,
PLA, |
Drug
delivery, devices |
crylate (PMMA) |
|
|
And PLGA |
|
|
|
|
|
|
Cellophane |
Dialysis |
|
|
|
|
membranes |
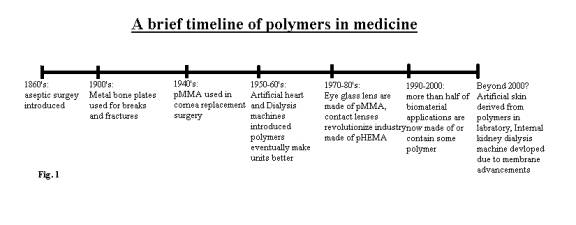
A brief history of polymers in medicine
The
inception of biomaterial was not seen until the 1860’s with the introduction of
aseptic surgical techniques3.
Although not practical for many years to come, the early 1900’s gave us
early uses of bone plates made from metals like steel to help mend breaks and
fractures. Metal alloys were
continuously the only form of biomaterials used for many years to come. Their applications ranged from joint
replacement to drug delivery systems.
Not
until World War II, would the rapidly developing science of polymers be seen in
medical applications.
Polymethylmethacrylate (pMMA) was one of the first polymers used as a
biomedical material serving as a material to replace the human cornea when
damaged3. Since then
scientists in the medical field have turned biomedical polymers into a billion
dollar business. Polymers with their
exceptional properties, their ability to engineered and their abundance in the
world have made a home in biomaterials and other medical applications.
Not
only have polymers replaced many old applications in medicine such as the shift
from metal catheters to those made of polyethylene, but also they have opened
the door for new applications that no other material would permit. Costly procedures have now been given new
lower cost alternatives. At one time a
severe heart condition called for an immediate transplant. In 1950’s the first artificial heart was
introduced. Brought in to practice in
the late 1960’s constant improvements, which include the uses of polyurethane’s
as ventricular assist devices, have not only made it safer to use in practice,
but the cost reduction has increased availability and helped the some 18,000
people a year who can not receive a donor immediately, a chance to wait a
little longer3.
What
once cost $480,000 and was only available to a few, now has a temporary
alternative that costs less than half that.
Polymers will continue to improve medicine and if the first fifty years
of development is any indication, the next fifty years will serve to save many
lives and help to make procedures and applications safer and more efficient.
Cellophane
Often
used in every day life to package our products or to keep our food fresh,
cellophane is one of the most critical materials for the treatment of many
kidney malfunctions. Cellophane is
regenerated cellulose. It is in the
form of a film; as opposed to rayon, which has the same properties yet is a
fiber. Cellophane holds almost
identical properties to the naturally occurring cellulose, it is “regenerated”
for processing purposes. It has a
typical chain length ranging from 2000 – 6000 angstroms (longer in fibers) and
a molecular weight of varying widely from 300,000 to one million g/mol4.
Cellophane
(regenerated Cellulose) was invented was invented by Jacques E. Brandenberger
in 1908. In an attempt to develop a
clear plastic cloth that was waterproof, he discovered that while his cloth
that he produced was to stiff, a clear plastic film would be peeled off and
soon known as cellophane5.
While years later processes would seal the permeability of this
regenerated cellulose and make it waterproof, the properties the original
material held would soon help save and prolong many lives.
In
1959 Dr. Willem J. Kolff’s first artificial kidney was installed in St. Paul’s
hospital in London. Ethical debate
would continue for 2 years, but in 1961 the first dialysis was performed. Within 5 years a separate unit was opened in
the hospital to treat patients suffering from renal problems. This machine used the idea of countercurrent
flow, osmosis and diffusion to remove waste products from the blood stream,
which are normally removed by the kidneys6.
The first
artificial kidney used vegetable parchment to serve as the separation membrane
between the fluids, which would selectively remove the undesirables. Natural casings (i.e. intestines) were also
used in the earlier stages of development of the artificial kidney. In the 1960’s Brandenberger’s original
cellophane was put to use as the membrane that filters and separates the
dialysis fluid from the blood. The
precursor to the saran wrap that we use today had properties that are so
desirable because of its ultra small permeability.
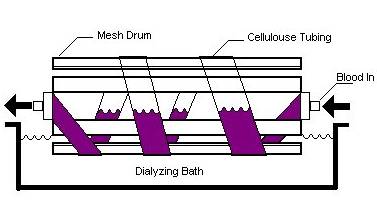
Fig. 2 A schematic of an artificial kidney
(hemodialysis)
Cellophane
membranes that separate the fluids in dialysis machines are expected to be
hydrophilic ultra filters, with the driving force being the concentration
gradient between the two fluids. Many
other properties are required between the fluids to make dialysis. Most
importantly the membrane permits many small particles in the blood with low
molecular weights such as inorganic salts, urea, and creatine pass through the
membrane while important components of the blood do not7.
The
production of cellophane as stated earlier is simply the regeneration of
cellulose. Obtained naturally from wood
and cotton fiber. Cellulose is reacted
with NaOH and carbon disulfide to produce cellulose xanthate. Cellulose xanthate is then treated with
sulfuric acid. The result of the
reaction is extruded to a sheet and after a small “aging” period, a thin clear
film of cellophane can be peeled. The
process by which cellophane is known is viscose4.
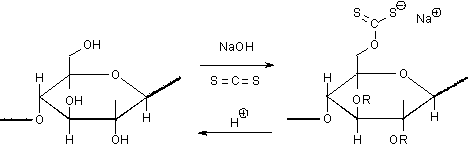
Fig. 3 The regeneration of Cellulose (cellophane).
While
early in history there were many concerns about potential health risks involved
with cleaning a person’s blood by a machine, debate quickly subsided with proof
of effectiveness. Today dialysis
machines save thousands of lives daily.
The only true alternative to dialysis is kidney transplant. Even in the very unlikely case of a
successful transplant (over 50% rejection rate) dialysis is continued for many
month or years, to ensure stability.
The
future of cellulose membranes in the treatment of renal failure has no
limits. Perhaps one-day membrane
efficiency will be so effective, predictable, and controllable enabling an
actual internal artificial kidney. The
main inconvenience with dialysis is the actual administration of the lengthy
procedure. With improvements in the
engineering of the membrane in dialysis, the result has been healthier patients
and longer lives for the unfortunate victims.
PGA, PLA,
and PLGA
The
polymer polyglycolic acid, PGA, initially started out as an absorbable suture
named Dexon. Dupont, under the direction of Norton Higgins, first synthesized
PGA by a three-step process from glycolic acid by manipulating temperature and
pressure. The ability of PGA to form biodegradable sutures, however, wasn’t
found until 1963 by Edward Schmitt and Rocco Polistina of the American Cyanamid
Corporation. Since the birth of PGA, derivatives of this polymer have been
found to have useful medical properties as well.
Modifying
the chemical and structural properties of PGA, PLA, and PLGA allows the
polymers to be used for a wide variety of applications within the human
body. These polymers are then used for
drug-delivery systems, to construct synthetic scaffolding, etc. The amorphous
form of PLA is used for drug delivery applications. The latest treatment in
treating brain tumors involves attaching dime-sized wafers directly into the
skull8. The wafers are made out of PLA or PLGA and slowly distribute
cancer-killing reagents directly into the location where it’s needed. The more
crystalline form of PLA has been found to useful as well. The mechanical
toughness and strength of the semi crystalline form of PLA and PGA is exploited
for use in orthopedic devices. Employing the polymers for the construction of
3-D scaffolding does this. The scaffolding is then implemented to grow new
tissues to replace damaged organs in the body.
All the polymers have very
low polydisperity index ratios, for example, the P.D.I. ratio for PLA is around
1.6-1.9. The low ratio is useful to maintain mechanical and structural
consistency for later applications. The most common method of commercial
production of PLA and PGA is by utilizing ring-opening polymerization combined
with an insertion mechanism using a metal oxide4. Depending on
structure, the polymers can be fitted for different applications. A more amorphous form of the polymer can be
used for drug delivery devices while the crystalline form is good for building
scaffolding and other biodegradable structures. PLGA, for example, is
completely amorphous so therefore it is used only in drug delivery devices. For
scaffolding, a more crystalline form of polymer is useful. Two essentials in
building scaffolding are having a high surface to volume ratio, and it has to
be highly porous. This is advantageous
since it allows to cells to easy proliferate and pathways for nutrients and
metabolites. The cells are first grown in a culture, and then are seeded onto
the scaffolding to grow the damaged organ9. The scaffolding gradually erodes away as
cells began to grow and replace lost tissue around the region. Using a lower
molecular weight polymer can speed up degradation.
PGA, PLA, and PGLA are new novel ways to
treat a variety of medical concerns.
There are some drawbacks, however, to their effectiveness. The use of
certain drugs, for example, is prohibited by the relatively temperatures used
in constructing these polymers. Another drawback is in the controlled release
of drugs. Bulk erosion has a somewhat inconsistent release of drugs. Depending
on the amount of drug loaded onto the polymer, the hydrophilic or hydrophobic
properties, the initial rate of release can vary. These persistent problems are
likely to be solved in the future. Due to recent legislation, which bars
suppliers of ingredients of medical components from being sued, researchers and
companies are freer to pursue medical applications and problems.
Polydimethyl siloxane
The
polymer polydimethyl siloxane (PDMS) is used in pacemakers, the delivery of
vaccines, and the construction cerebrospinal fluid shunts. PDMS is an amorphous
structure with low cross-linked elasticity.
As a vulcanized rubber it cannot be melted or dissolved. The glass transition temperature of PDMS is
very low (150K), and the polymer is very permeable to gases. The low glass transition temperature allows
for fast molecular relaxation, which is beneficial for molding applications.
An
English chemist, Dr. Frederic Stanley Kipping, discovered silioxanes in
1927. Kipping however, incorrectly
analyzed the structure of his newly found macromolecule and as a result he
called his discovery silicone. This name still persists today. It wasn’t until 1943, however, that mass
production silicones occurred. General Electric started industrial production
under the direction of Eugen Rochow.
PDMS
are used in numerous beneficial applications.
For example, PDMS became an essential ingredient for use in glass eyes
in World War 2. Prior to the inception
of localized drug delivery within the human body, antigens had to be taken
orally and it was difficult, if not impossible to simulate local immune
response in the body.
This principle of localized drug delivery
using PDMS comes into play in radical prostatectomy and radiation therapy for
treatment of prostate carcinoma. There are several complications due to the
surgery “ the most significant complication is post operative incontinence,
which affects 30% of patients”10.
Since PDMS stays localized in the injection site a lesser dosage of
drugs is needed due to the increased concentration in the affected area. For
the delivery of the vaccine, biodegradable pellets made of PDMS are used. The
pellets are very small in diameter and generally contain soluble antigens to be
released within the body. The pellets consist of vulcanized rubber and have a
mean diameter of 188 um which allows for the particles to stay in the localized
region. Drug release is controlled “ by
the relative magnitude of the velocity of macromolecular relaxation to the
velocity of drug diffusion through the rubbery region.”11. Also PMS
isn’t very susceptible to bacterial infection. This property also makes it ideal for use in pacemakers and the
construction of cerebrospinal fluid shunts where the chance for cancer becomes
nil.
One method
for the production of dimethyl siloxane starts with the monomer,
dichlorodimethylsilane. Hydroxyl groups, through hydrolysis, replace the two
chlorines in the monomer. To achieve a higher molecular weight, however, a
different approach is used. This new method is done by a “ base catalyzed
ring-opening polymerization of the siloxanes.”4. Most major
producers of PDMS aren’t involved in the medical industry. PDMS is mainly found
in worldly applications such as lubricants, foaming agents, etc.
The main
public concern for the use of PDMS stems from post-operative complications.
Troubles in surgery usually start after the implanted device becomes
contaminated with microorganisms or the wound becomes infected. Even under the
most stringent antiseptics conditions, contamination is still a factor that has
to be taken into account. Bacterial infection at the site of the catheter could
occur for several reasons including surface adhesion and growth, production of
extracellular components (slime), etc.
PDMS, however, is still at the forefront of
medical research, whose novel properties warrants further research.
Polyethylene
and Polymethylmethacrylate (PMMA)
Many common thermoplastics,
such as polyethylene and polyester, are used as biomaterials. Thermoplastics
usually exhibit moderate to high tensile strength (5 to 1,000 megapascals) with
moderate elongation (2 to 100 percent), and they undergo plastic deformation at
high strains. Depending on the structure and molecular organization of the
polymer chains, thermoplastics may be
semi-crystalline or highly crystalline.
Joint replacements, particularity at the
hip, and bone fixation devices have become very successful applications of
materials in medicine. The use of pins, plates, and screws for bone fixation to
aid recovery of bone fractures has become routine, with the number of annual
procedures approaching five million in the USA alone12.
Hip-joint replacements are principally
used for structural support. Consequently, materials that possess high
strength, such as metals, tough plastics, and reinforced polymer-matrix
composites dominate them. In addition, biomaterials used for orthopedic
applications must have high modulus, long-term dimensional stability, high
fatigue resistance, and biocompatibility(i.e., there should be no adverse
tissue response to the implanted device). Early developments in this field used
readily available materials such as stainless steels, but evidence of corrosion
after implantation led to their replacement by more stable materials,
particularly titanium alloys, cobalt-chromium-molybdenum alloys, and carbon
fiber-reinforced polymer composites. A typical modern artificial hip consists
of a nitrided and highly polished cobalt-chromium ball connected to a titanium
alloy stem that is inserted into the femur and cemented into place by in situ
polymerization of polymethylmethacrylate.
Consequently, much research on the
development of hip-joint materials has been devoted to optimizing the
properties of the articulating components in order to eliminate surface wear.
Other modifications include porous coatings made by sintering the metal surface
or coatings of wire mesh or hydroxyapatite; these promote bone growth and
integration between the implant and the host, eliminating the need for acrylic
bone cement13.
Polytetrafluoroethylene
PTFE is thermosetting polymer very limited application in medicine, but
its characteristic properties, which combine high strength and chemical
resistance, are useful for some orthopedic and dental devices. It also has high
modulus and tensile properties with negligible elongation. The polymer chains
in this material are highly cross-linked and therefore have severely macromolecular
mobility; this limits extension of the polymer chains under an applied
load.
Biomaterials are used in many blood-contacting devices. These include
artificial heart valves, synthetic vascular grafts, ventricular assist devices,
drug releases, and a wide range of invasive treatment and diagnostic systems.
An important issue in the design and selection of materials is the hemodynamic
conditions in the vicinity of the device. For instance, mechanical heart
valve implants are intended for long-term use. Consequently, the hinge
points of each valve leaflet and the materials must have excellent wear and
fatigue resistance in order to open and close 80 times per minute for many
years after implantation. In addition, the open valve must minimize disturbances
to blood flow as blood passes from the left ventricle of the heart, through the
valve and into the ascending aorta of the arterial vascular system. To this
end, the bileaflet valve disks of one type of implant are coated with pyrolytic
carbon, which provides a relatively smooth, chemically inert surface.
Synthetic vascular graft materials are used to patch injured or diseased
areas of arteries, for replacement of whole segments of larger arteries such as
the aorta, and for use as sewing cuffs. Such materials need to be flexible to
allow for the difficulties of implantation and to avoid irritating adjacent
tissues; also, the internal diameter of the graft should remain constant under
a wide range of flexing and bending conditions, and the modulus or compliance
of the vessel should be similar to that of the natural vessel. A biomaterial
used for blood vessel replacement will be in contact not only with blood but
also with adjacent soft tissue. Experience with different materials has shown
that tissue growth into the interstices of the biomaterials aids healing and
integration of the material with host tissue after implantation. In order for
the tissue, which consists mostly of collagen, to grow in the graft, the
vascular graft must have an open structure with pores at least 10 micrometers
in diameter. Fibroblasts synthesize the structural protein tropocollagen, which
is needed in the development of new fibrous tissue as part of the healing
response to a surgical wound.
Artificial heart valves and vascular grafts, while not ideal, have been
used successful and have saved many thousands of lives. However, the risk of
thrombosis has limited the success of existing cardiovascular devices and has
restricted potential application of the biomaterials to other devices.
Considerable advances have been made in the ability to manipulate molecular
architecture at the surface of materials by using chemisorbed or physisorbed
monolayer films. Such progress in surface modification, combined with the
development of nanoscale probes that permit examination at the molecular and
submolecular level, provide a strong basis for optimism in the development of
specialty biomaterials with improved blood compatibility14.
Polyurethane
Seen
today in everyday uses such as shoe soles, tires and foams, polyurethane holds
an extremely import role in cardiac medicine today. Polyurethane is a thermoset that is also a non-condensation step
growth polymer4.
Polyurethane has a very low molecular weight compared to many other polymers
with a molecular weight average of only 47,000 g/mol. The benefits of this material lie in the basics of it visible
physical properties. Polyurethane is
often described to bridge the gap between rubber and plastic. It holds one of the best load-bearing
capacities of almost any materials around15.
Invented
back in 1937 by Otto Baker, polyurethane was the result of a search for a
material that has high strength and good environmental resistance. For both reasons polyurethane today is one
of the most important materials in use for ventricular assist devices. Differing from artificial hearts, VAD’s are
for short-term assistance to cardiac circulation attached to one or both of the
heart ventricles. Most commonly seen in
the operating room during open-heart surgery, postoperatively, and in the cases
of extreme cardiac trauma. They consist
of tubing attached to the heart valves leading to a pump that can be
centrifugal, electrical, or pneumatic.
While
Dr. D. Liotta of Baylor University developed the principles of this device in
the 1950’s, two doctors, Pierce and Donachy in 1971, significantly refined the
technology. Rewriting the book on fluid
mechanics (relating to blood flow) and taking advantage of polymers as a
material. Polyurethane (segmented)
stabilized the VAD, making not only the contact barrier of the blood and
machine the safest possible, but also using the compressive properties that it
exhibits made it function more like the actual heart itself. The once majority metal device was revealed
in 1976 and approved for use by the FDA in 198016.
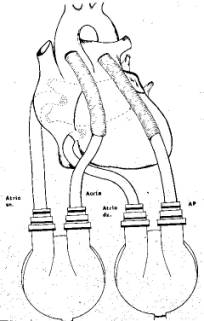
Fig. 4 Schematic of a Ventricular Assist
Device
There
are two ways to produce polyurethane.
However, the most abundant source (95% of world production) is obtained
through step growth polymerization of diisocyanates with dihydroxl
compounds. The result is a polymer that
has a load bearing capacity comparable to cast steel. Polyurethane is molded most often through injection molding. Additionally as of recent years, reaction
injection molding (RIM) has become one of the more popular ways to produce in
industry. For the most polyurethane
used for VAD’s are produced under careful supervision and not often RIM
produced. The largest debate over the
use of these materials was potential for mechanical failure. Past occurrences of failure have been
attributed to poor processing and not the material itself17.
VAD’s
have made great strides in the past 20 years.
Where once limited to external unit only, today there are internally
placed VAD’s and as the technology improves, it may one day replace transplant
surgery to cure cardiac conditions.
While there are many types of VAD’s, the only material that is a true
alternative in some sense to the polyurethane is stainless steel. Advancements in the use of ventricular
assist devices can be seen in the decrease in number of deaths of patients
awaiting transplant, even with a great increase of people on the waiting
list. Perhaps not a solution, the
temporary alternative that exists in VAD’s can only be attributed the
integration of polymers into their design.
Conclusion
Indeed, biomaterials
have already made a huge impact on medical practices. But, the opportunities
that lie ahead of us are enormous. “Tissue engineering and related subjects
have the potential to change paradigms” for treating diseases that today cannot
be treated effectively like certain forms of liver failure, paralysis, and
certain disorders. “ Clearly we are faced with big challenges “. But, the
message I try to get across to everyone mostly to young students is that the
field holds a tremendous promise1.
References
1. Peppas, N., Langer, R. “New challenges in bio-materials”, Science, Vol. 263, March, 1994
2.
Andreadis, S., “Applications of Biomaterials”,
Tissue engineering handout, February 2001, University at Buffalo.
3.
“History and Development of Biomaterials”, www.bae.ncsu.edu/Courses/bae465
4.
Fried, J. R., “Polymer Science and Technology.”, Prentice Hall,
New Jersey 1995
5.
“Cellophane Invention”,
http://inventors.about.com/science/inventors/library/inventors/blcellophane.htm
6.
“First
Dialysis Unit”, www.ucl.ac.uk/uro-neph/history/dialysis.htm
7.
“Dialysis
and the Artificial Kidney”, www.chemengineer.about.com/science/chemengineer/library/weekly/aa120897.htm
9.
Ikada, Y, Yoshihiko, S, “Tissue Engineering for Therapeutic Use
4.” Elsevier, 2000, New York
10.
Pulverer, G., Schierholz, J. M., “Development of New CSF-shunt
With Sustained Release of Antimicrobial Broad-Spectrum Combination.”,
Baktercologie, Vol. 286, 107-123
11.
Loomes, L. M., Jian Xiong, J., Brook, M. A., Underdown, B. J.,
McDermott, M. R., “Novel Polymer-grafted Starch Microparticles for Mucosal
Delivery of Vaccines.”, Immunology, Vol. 56, 162-168, 1996
12.
www.britannica.com,
(keyword “polyethylene”)
13.
“Uses of Polymehtylmethacrylate”, www.rcsed.ac.uk (Feb 2001)
14.
www.britannica.com,
(keyword “Polytetrafluoroethylene”)
15.
“Polyurethane – Features and Benefits”, www.elastchem-ca.com/poly.html
16.
“Pierce-Donachy Ventricular Assist Device”, www.asme.org/history/Roster/H142.html
17.
Liotta, D. “The Ventricular Assist Device”, www.fdliotta.org